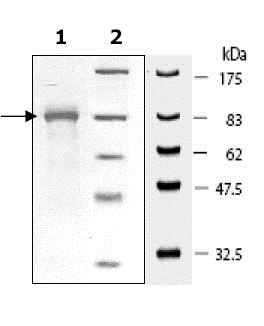
Recombinant Human Hsp90 beta protein (ab80353)
Key features and details
- Expression system: Escherichia coli
- Purity: > 95% SDS-PAGE
- Tags: His tag C-Terminus
- Suitable for: SDS-PAGE
Description
-
Product name
Recombinant Human Hsp90 beta protein
See all Hsp90 beta proteins and peptides -
Purity
> 95 % SDS-PAGE. -
Expression system
Escherichia coli -
Protein length
Full length protein -
Animal free
No -
Nature
Recombinant -
-
Species
Human -
Tags
His tag C-Terminus
-
Associated products
-
Related Products
Specifications
Our Abpromise guarantee covers the use of ab80353 in the following tested applications.
The application notes include recommended starting dilutions; optimal dilutions/concentrations should be determined by the end user.
-
Applications
SDS-PAGE
-
Form
Liquid -
 Concentration information loading...
Concentration information loading...
Preparation and Storage
-
Stability and Storage
Shipped on Dry Ice. Upon delivery aliquot. Store at -80°C. Avoid freeze / thaw cycle.
pH: 8
Constituents: 0.0462% (R*,R*)-1,4-Dimercaptobutan-2,3-diol, 0.395% Tris HCl, 0.05% Tween, 20% Glycerol (glycerin, glycerine), 0.58% Sodium chloride
General Info
-
Alternative names
- 90 kda heat shock protein beta HSP90 beta
- D6S182
- FLJ26984
see all -
Function
Molecular chaperone that promotes the maturation, structural maintenance and proper regulation of specific target proteins involved for instance in cell cycle control and signal transduction. Undergoes a functional cycle that is linked to its ATPase activity. This cycle probably induces conformational changes in the client proteins, thereby causing their activation. Interacts dynamically with various co-chaperones that modulate its substrate recognition, ATPase cycle and chaperone function. -
Sequence similarities
Belongs to the heat shock protein 90 family. -
Domain
The TPR repeat-binding motif mediates interaction with TPR repeat-containing proteins. -
Post-translational
modificationsUbiquitinated in the presence of STUB1-UBE2D1 complex (in vitro).
ISGylated.
S-nitrosylated; negatively regulates the ATPase activity. -
Cellular localization
Cytoplasm. Melanosome. Identified by mass spectrometry in melanosome fractions from stage I to stage IV. - Information by UniProt
Images
Protocols
To our knowledge, customised protocols are not required for this product. Please try the standard protocols listed below and let us know how you get on.
Datasheets and documents
-
SDS download
-
Datasheet download
References (2)
ab80353 has been referenced in 2 publications.
- Christensen B et al. Erythropoietin does not activate erythropoietin receptor signaling or lipolytic pathways in human subcutaneous white adipose tissue in vivo. Lipids Health Dis 15:160 (2016). WB ; Human . PubMed: 27640183
- Labrador-Garrido A et al. Chaperome screening leads to identification of Grp94/Gp96 and FKBP4/52 as modulators of the a-synuclein-elicited immune response. FASEB J 30:564-77 (2016). PubMed: 26443817