Anti-alpha A Crystallin/CRYAA antibody (ab139503)
Key features and details
- Rabbit polyclonal to alpha A Crystallin/CRYAA
- Suitable for: WB
- Reacts with: Human
- Isotype: IgG
Get better batch-to-batch reproducibility with a recombinant antibody
- Research with confidence – consistent and reproducible results with every batch
- Long-term and scalable supply – powered by recombinant technology for fast production
- Success from the first experiment – confirmed specificity through extensive validation
- Ethical standards compliant – production is animal-free
Overview
-
Product name
Anti-alpha A Crystallin/CRYAA antibody
See all alpha A Crystallin/CRYAA primary antibodies -
Description
Rabbit polyclonal to alpha A Crystallin/CRYAA -
Host species
Rabbit -
Tested applications
Suitable for: WBmore details -
Species reactivity
Reacts with: Human
Predicted to work with: Mouse, Rat, Chimpanzee, Gorilla, Orangutan
-
Immunogen
Synthetic peptide corresponding to Human alpha A Crystallin/CRYAA aa 150 to the C-terminus (C terminal) conjugated to keyhole limpet haemocyanin.
Database link: P02489 -
Positive control
- This antibody gave a positive signal in Human Brain and Spinal Cord tissue lysates.
-
General notes
The Life Science industry has been in the grips of a reproducibility crisis for a number of years. Abcam is leading the way in addressing this with our range of recombinant monoclonal antibodies and knockout edited cell lines for gold-standard validation. Please check that this product meets your needs before purchasing.
If you have any questions, special requirements or concerns, please send us an inquiry and/or contact our Support team ahead of purchase. Recommended alternatives for this product can be found below, along with publications, customer reviews and Q&As
Properties
-
Form
Liquid -
Storage instructions
Shipped at 4°C. Store at +4°C short term (1-2 weeks). Upon delivery aliquot. Store at -20°C or -80°C. Avoid freeze / thaw cycle. -
Storage buffer
pH: 7.40
Preservative: 0.02% Sodium azide
Constituent: PBS
Batches of this product that have a concentration < 1mg/ml may have BSA added as a stabilising agent. If you would like information about the formulation of a specific lot, please contact our scientific support team who will be happy to help. -
 Concentration information loading...
Concentration information loading... -
Purity
Immunogen affinity purified -
Clonality
Polyclonal -
Isotype
IgG -
Research areas
Associated products
-
Compatible Secondaries
-
Isotype control
-
Recombinant Protein
Applications
The Abpromise guarantee
Our Abpromise guarantee covers the use of ab139503 in the following tested applications.
The application notes include recommended starting dilutions; optimal dilutions/concentrations should be determined by the end user.
| Application | Abreviews | Notes |
|---|---|---|
| WB |
Use a concentration of 1 µg/ml. Detects a band of approximately 20 kDa (predicted molecular weight: 20 kDa).
|
| Notes |
|---|
|
WB
Use a concentration of 1 µg/ml. Detects a band of approximately 20 kDa (predicted molecular weight: 20 kDa). |
Target
-
Function
May contribute to the transparency and refractive index of the lens. -
Involvement in disease
Defects in CRYAA are a cause of cataract autosomal dominant (ADC) [MIM:604219]. Cataract is an opacification of the crystalline lens of the eye that frequently results in visual impairment or blindness. Opacities vary in morphology, are often confined to a portion of the lens, and may be static or progressive. In general, the more posteriorly located and dense an opacity, the greater the impact on visual function. Cataract is the most common treatable cause of visual disability in childhood. -
Sequence similarities
Belongs to the small heat shock protein (HSP20) family. -
Post-translational
modificationsO-glycosylated; contains N-acetylglucosamine side chains.
Deamidation of Asn-101 in lens occurs mostly during the first 30 years of age, followed by a small additional amount of deamidation (approximately 5%) during the next approximately 38 years, resulting in a maximum of approximately 50% deamidation during the lifetime of the individual.
Phosphorylation on Ser-122 seems to be developmentally regulated. Absent in the first months of life, it appears during the first 12 years of human lifetime. The relative amount of phosphorylated form versus unphosphorylated form does not change over the lifetime of the individual. -
Cellular localization
Cytoplasm. Nucleus. Translocates to the nucleus during heat shock and resides in sub-nuclear structures known as SC35 speckles or nuclear splicing speckles. - Information by UniProt
-
Database links
- Entrez Gene: 1409 Human
- Entrez Gene: 12954 Mouse
- Entrez Gene: 24273 Rat
- Omim: 123580 Human
- SwissProt: P02489 Human
- SwissProt: P24622 Mouse
- SwissProt: P24623 Rat
- Unigene: 184085 Human
see all -
Alternative names
- Acry 1 antibody
- Alpha crystallin A chain antibody
- Alpha-crystallin A chain antibody
see all
Images
-
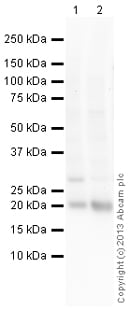
All lanes : Anti-alpha A Crystallin/CRYAA antibody (ab139503) at 1 µg/ml
Lane 1 : Brain (Human) Tissue Lysate - adult normal tissue
Lane 2 : Spinal Cord (Human) Tissue Lysate - adult normal tissue
Lysates/proteins at 10 µg per lane.
Secondary
All lanes : Donkey Anti-Rabbit IgG H&L preadsorbed (ab97081) at 1/10000 dilution
Developed using the ECL technique.
Performed under reducing conditions.
Predicted band size: 20 kDa
Observed band size: 20 kDa
Additional bands at: 28 kDa (possible non-specific binding)
Exposure time: 30 secondsThis blot was produced using a 4-12% Bis-tris gel under the MES buffer system. The gel was run at 200V for 35 minutes before being transferred onto a Nitrocellulose membrane at 30V for 70 minutes. The membrane was then blocked for an hour using 5% Bovine Serum Albumin before being incubated with ab139503 overnight at 4°C. Antibody binding was detected using an anti-rabbit antibody conjugated to HRP, and visualised using ECL development solution.
Datasheets and documents
-
SDS download
-
Datasheet download
References (0)
ab139503 has not yet been referenced specifically in any publications.