Atto 488 Conjugation Kit (Fast) - Lightning-Link® (ab269896)
Overview
-
Product name
Atto 488 Conjugation Kit (Fast) - Lightning-Link® -
Product overview
Atto 488 Conjugation Kit / Atto 488 Labeling Kit (ab269896) uses a simple and quick process for Atto 488 labeling / conjugation of antibodies. It can also be used to conjugate other proteins or peptides. Learn about our antibody labeling kits and their advantages.
To conjugate an antibody to Atto 488 using this kit:
- add modifier to antibody and incubate for 15 mins
- add quencher and incubate for 5 mins
The Atto 488 conjugated antibody can be used immediately in WB, ELISA, IHC etc. No further purification is required and 100% of the antibody is recovered for use.
The excitation and emmision wavelengths for Atto 488 are Ex: 500nm, Em: 520nm.
Learn about buffer compatibility below; for incompatible buffers and low antibody concentrations, use our rapid antibody purification and concentration kits. Use the FAQ to learn more about the technology, or about conjugating other proteins and peptides to Atto 488.
Custom size conjugation kits up to 100 mg are available on demand. Please contact us to discuss your requirements.
-
Notes
This product is manufactured by Expedeon, an Abcam company, and was previously called Lightning-Link® Rapid Atto 488 Labeling Kit. 350-0015 is the same as the 1 mg size. 350-0010 is the same as the 3 x 100 ug size. 350-0030 is the same as the 3 x 10 ug size. 350-0005 is the same as the 100 µg size.
Amount and volume of antibody for conjugation to Atto 488
Kit size Recommended
amount of antibody1Maximum
amount of antibodyMaximum antibody
volume23 x 10 µg 3 x 10 µg 3 x 20 µg 3 x 10 µL 100 µg 100 µg 200 µg 100 µL 3 x 100 µg 3 x 100 µg 3 x 200 µg 3 x 100 µL 1 mg 1 x 1 mg 1 x 2 mg 1 x 1 mL 1 Using the maximum amount of antibody may result in less labeling per antibody.
2 Ideal antibody concentration is 1mg/ml. 0.5 - 1 mg/ml can be used if the maximum antibody volume is not exceeded. Antibodies > 2mg/ml or < 0.5 mg/ml should be diluted /concentrated.
Buffer Requirements for Conjugation
Buffer should be pH 6.5-8.5.
Compatible buffer constituents
If a concentration is shown, then the constituent should be no more than the concentration shown. If several constituents are close to the limit of acceptable concentration, then this can inhibit conjugation.50mM / 0.6% Tris1 0.1% BSA2 50% glycerol 0.1% sodium azide PBS Potassium phosphate Sodium chloride HEPES Sucrose Sodium citrate EDTA Trehalose 1 Tris buffered saline is almost always ≤ 50 mM / 0.6%
2 BSA can also interfere with the use of the conjugated antibody in tissue staining.Incompatible buffer constituents
Thiomerosal Proclin Glycine Arginine Glutathione DTT If a constituent of the buffer containing your antibody or protein is not listed above, please check the FAQ or contact us.
Only purified antibodies are suitable for use, ie. where other proteins, peptides, or amino acids are not present: antibodies in ascites fluid, serum or hybridoma culture media are incompatible.
Storing and handling conjugation kits
Lyophilized Lightning-Link® components are hygroscopic.
Kits are intentionally shipped at ambient temperature with silica gel to avoid exposure to moisture. Upon receipt, store the kit frozen and protect from moisture. Before opening the outer container, allow the lyophilized components to reach room temperature to minimize condensation.
Properties
-
Storage instructions
Store at -20°C. Please refer to protocols. -
Components 100 µg 1 mg 3 x 10 µg 3 x 100 µg ab274065 - Atto 488 mix (Lyophilized) 1 x 100µg 1 x 1mg 3 x 10µg 3 x 100µg ab273994 - Modifier reagent 1 x 200µl 1 x 200µl 1 x 200µl 1 x 200µl ab273995 - Quencher reagent 1 x 200µl 1 x 200µl 1 x 200µl 1 x 200µl
Images
-
 Immunocytochemistry - Atto 488 Conjugation Kit (Fast) - Lightning-Link (ab269896)Image from Eichwald, Catherine, et al., PloS one; 7(10): e47947. doi: 10.1371/journal.pone.0047947. Reproduced under the Creative Commons license https://creativecommons.org/licenses/by/4.0/
Immunocytochemistry - Atto 488 Conjugation Kit (Fast) - Lightning-Link (ab269896)Image from Eichwald, Catherine, et al., PloS one; 7(10): e47947. doi: 10.1371/journal.pone.0047947. Reproduced under the Creative Commons license https://creativecommons.org/licenses/by/4.0/Eichwald, Catherine, et al used Atto 488 Conjugation Kit (Fast) - Lightning-Link® (ab269896) as part of examining the distribution of acetylated-microtubules (MTs) with respect to viroplasms. They used the kit to conjugate Atto 488 to mouse monoclonal anti-alpha tubulin antibody for use in immunocytochemistry/immunofluorescence (ICC/IF).
At 6 hours post-infection, SA11-infected CV-1 cells [multiplicity of infection (MOI); 25 VFU/ml] were fixed with methanol and immunostained for detection of viroplasms in red (anti-NSP5 antibody followed by a secondary antibody conjugated to Alexa 594), acetylated-MTs in cyan (mouse mAb anti-acetylated alpha-tubulin followed by a secondary antibody conjugated to Alexa 647), MTs in green (mouse mAb anti-alpha tubulin directly conjugated to Atto 488 (green)) and nuclei in blue (DAPI). Z-stack images were acquired by high-resolution CLSM and subsequently, 3D-reconstructions were performed using surface and filament algorithms from the surpass model of the Imaris 7.0 software (Bitplane, Switzerland). 3D-reconstructions are visualized from the topside (A, B and C) and the bottom side (D, E and F) of the preparation. Images show viroplasms (from A to F), acetylated-MTs (A, B, D and E) and MTs (A, C, D and F). Scale bars are 5 µm. -
 Immunocytochemistry - Atto 488 Conjugation Kit (Fast) - Lightning-Link(ab269896)Image from Simanjuntak, Yogy, et al., Frontiers in microbiology 8:651; doi: 10.3389/fmicb.2017.00651. Reproduced under the Creative Commons license https://creativecommons.org/licenses/by/4.0/
Immunocytochemistry - Atto 488 Conjugation Kit (Fast) - Lightning-Link(ab269896)Image from Simanjuntak, Yogy, et al., Frontiers in microbiology 8:651; doi: 10.3389/fmicb.2017.00651. Reproduced under the Creative Commons license https://creativecommons.org/licenses/by/4.0/Simanjuntak, Yogy, et al used Atto 488 Conjugation Kit (Fast) - Lightning-Link® (ab269896) as part of examining the mechanisms of viral invasiveness in Japanese encephalitis virus (JEV). They used the kit to conjugate Atto 488 to Japanese encephalitis virus (JEV) for use in immunocytochemistry.
BE(2)C cells were infected with fluorescently labeled JEV (Atto488) for 2 h at room temperature with or without anti-integrin β3 and anti-vimentin antibody (0.75 and 1.5 μg/ ml). Mouse immunoglobulin G (IgG) was used as antibody control (1.5 μg/ml). Cells with or without antibody (1.5 μg/ ml) were stained with Alexa Fluor 568 phalloidin for F-actin (red) and Hoechst for nuclei (blue). Confocal microscopy images (630× original magnification). -
 Immunohistochemistry (Frozen sections) - Atto 488 Conjugation Kit (Fast) - Lightning-Link(ab269896)Image from Veldkamp, Marieke W., et al., Nature communications, 9(1):4357. doi: 10.1038/s41467-018-06530-5. Reproduced under the Creative Commons license https://creativecommons.org/licenses/by/4.0/
Immunohistochemistry (Frozen sections) - Atto 488 Conjugation Kit (Fast) - Lightning-Link(ab269896)Image from Veldkamp, Marieke W., et al., Nature communications, 9(1):4357. doi: 10.1038/s41467-018-06530-5. Reproduced under the Creative Commons license https://creativecommons.org/licenses/by/4.0/Veldkamp, Marieke W., et al used Atto 488 Conjugation Kit (Fast) - Lightning-Link® (ab269896) as part of examining electrophysiological effects of the neuropeptide Sub-P in isolated rabbit atrial myocytes, and in Langendorff-perfused and in situ rabbit heart. They used the kit to conjugate Atto 488 to anti-NK3R antibody for use in immunohistochemistry (frozen sections).
Sections of rabbit (upper panel) and human (lower panel) left atrium showing labelling of the NK-3R (green) in the peripheral sarcolemma of cardiomyocytes, co-stained for the intracellular myocardial marker a-actinin (red). Scale bar 25 µM. -
 Flow Cytometry - Atto 488 Conjugation Kit (Fast) - Lightning-LinkImage from Robinson, Andrew P., et al. PloS One, 9(9): e107649.; doi: 10.1371/journal.pone.0107649. Reproduced under the Creative Commons license https://creativecommons.org/licenses/by/4.0/
Flow Cytometry - Atto 488 Conjugation Kit (Fast) - Lightning-LinkImage from Robinson, Andrew P., et al. PloS One, 9(9): e107649.; doi: 10.1371/journal.pone.0107649. Reproduced under the Creative Commons license https://creativecommons.org/licenses/by/4.0/Robinson, Andrew P., et al used Atto 488 Conjugation Kit (Fast) - Lightning-Link® (ab269896) as part of characterizing oligodendroglial populations. They used the kit to conjugate Atto 488 to Rat anti-PDGFRalpha antibody, clone APA5, for use in flow cytometry.
SJL/J mice were immunized with PLP139–151 and scored daily for clinical disease. A cohort of SJL/J mice was sacrificed, and spinal cords were analyzed by flow cytometry (n = 5). (A) Cells were distinguished from debris by forward and side scatter then singlet cells were gated. Live cells were gated by dead cell exclusion, and CNS resident cells were identified as CD45− or CD45low. (B) Oligodendroglial cells were defined by double positive staining: A2B5+PDGFRα+ early OPCs, A2B5+NG2+ intermediate OPCs, NG2+O4+ late OPCs, O4+MOG+ pre-myelinating oligodendrocytes, and GALC+MOG+ mature oligodendrocytes. -
 Atto 488 Conjugation Kit (Fast) - Lightning-Link® labeling TcdA1–1874 toxin for Flow cytometryImage from Olling A et al., PLoS One, 6(3):e17623. Fig 5.; doi: 10.1371/journal.pone.0017623. Reproduced under the Creative Commons license https://creativecommons.org/licenses/by/4.0/
Atto 488 Conjugation Kit (Fast) - Lightning-Link® labeling TcdA1–1874 toxin for Flow cytometryImage from Olling A et al., PLoS One, 6(3):e17623. Fig 5.; doi: 10.1371/journal.pone.0017623. Reproduced under the Creative Commons license https://creativecommons.org/licenses/by/4.0/Olling A et al. used ab269896 as part of examining the role of toxins in the pathogenicity of Clostridium difficile.
They used the kit to conjugate Atto 488 to TcdA1–1874 toxin for use in flow cytometry.
Binding of fluorescent labeled TcdA-PE/Cy5 and TcdA1–1874-Atto488 to HT29 and CHO-C6 cells was investigated by FACS analysis. Right shift of the black curve illustrates toxin binding which was detected through fluorescence emission at 667 nm for TcdA and at 523 nm for TcdA1–1874, respectively. Due to different ratio of fluorophor and toxin, fluorescence intensity of TcdA-PE/Cy5 cannot directly be compared with TcdA1–1874-Atto488.
-
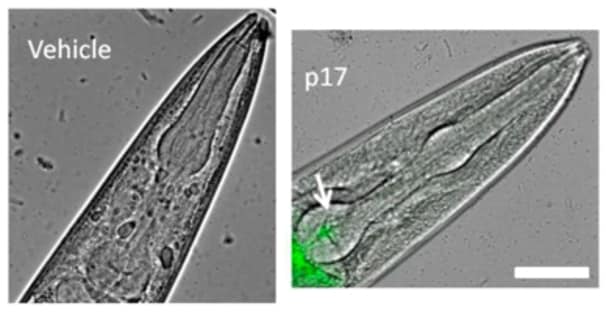 Atto 488 Conjugation Kit (Fast) - Lightning-Link® labeling p17 protein for MicroscopyImage from Zeinolabediny Y et al., Sci Rep., 7(1):10313. Fig 3.; doi: 10.1038/s41598-017-10875-0. Reproduced under the Creative Commons license https://creativecommons.org/licenses/by/4.0/
Atto 488 Conjugation Kit (Fast) - Lightning-Link® labeling p17 protein for MicroscopyImage from Zeinolabediny Y et al., Sci Rep., 7(1):10313. Fig 3.; doi: 10.1038/s41598-017-10875-0. Reproduced under the Creative Commons license https://creativecommons.org/licenses/by/4.0/Zeinolabediny Y et al. used ab269896 as part of examining the toxicity of p17 protein assemblies.
They used the kit to conjugate Atto 488 to p17 protein for use in microscopy.
Representative images of the p17 localisation as overlays of Atto 488-fluorescence and light microscopy in worms.
Scale bar = 50 µm. Worms fed for 2 h vehicle (10 mM PB, pH 7.4) or 4 nM p17-Atto 488.
Protocols
Datasheets and documents
-
SDS download
-
Datasheet download
References (20)
ab269896 has been referenced in 20 publications.
- Bian J et al. Limited AT1 Receptor Internalization Is a Novel Mechanism Underlying Sustained Vasoconstriction Induced by AT1 Receptor Autoantibody From Preeclampsia. J Am Heart Assoc 8:e011179 (2019). PubMed: 30845870
- Veldkamp MW et al. Neurokinin-3 receptor activation selectively prolongs atrial refractoriness by inhibition of a background K+ channel. Nat Commun 9:4357 (2018). PubMed: 30341287
- Carignan D et al. Activation of innate immunity in primary human cells using a plant virus derived nanoparticle TLR7/8 agonist. Nanomedicine 14:2317-2327 (2018). PubMed: 29128662
- Lindner K et al. Low Dose Carbon Black Nanoparticle Exposure Does Not Aggravate Allergic Airway Inflammation in Mice Irrespective of the Presence of Surface Polycyclic Aromatic Hydrocarbons. Nanomaterials (Basel) 8:N/A (2018). PubMed: 29614747
- Chen CY et al. Suppression of detyrosinated microtubules improves cardiomyocyte function in human heart failure. Nat Med 24:1225-1233 (2018). PubMed: 29892068




